RESEARCH
Understanding how cells grow and divide has profound impacts on basic science, biotechnology, and medicine. Despite recent advances in molecular biology and biochemistry, a central challenge remains: bridging the nanometer-scale activities of proteins and the construction of entire cells. Although the mechanisms of bacterial proliferation have been a major focus of research for over a century, it has remained difficult to determine how cellular structure and organization are dynamically controlled due to the central—yet neglected—importance of physical factors.
To address these knowledge gaps, our lab pursues research directions that span from the atomic to the multicellular scales. We investigate the physical nature of intracellular spatial organization, mechanics, and kinetics by leveraging top-down approaches based on cellular-scale observations, bottom-up approaches based on biophysical molecular observations, and computational modeling that connects the two paradigms. Understanding cellular growth and form remains a fascinating, multifaceted challenge with obvious implications for health and disease. In addition to the importance of bacteria as a model system for basic science, uncovering the general physical rules that underlie how bacteria grow and divide will have important applications for controlling bacterial communities and developing novel strategies in synthetic biology.
| Research Topics |
|---|
| The evolution of cell size and shape |
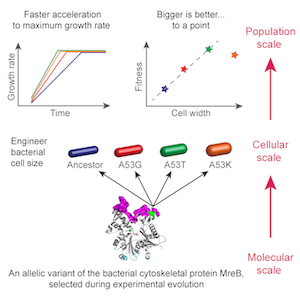 To examine the relationship between cell geometry and fitness, we systematically engineered strains with mreB point mutations that confer a range of changes in cell width. Fitness increases linearly as a function of cell width, indicating that changes in cell geometry are a direct cause of fitness increases (Monds et al., 2014). Motivated by this work, we have developed an experimental strategy to precisely tune cell size over a wide range, allowing us to separately measure the connections among cell geometry, growth, and size, eventually in a variety of organisms and shapes.
To examine the relationship between cell geometry and fitness, we systematically engineered strains with mreB point mutations that confer a range of changes in cell width. Fitness increases linearly as a function of cell width, indicating that changes in cell geometry are a direct cause of fitness increases (Monds et al., 2014). Motivated by this work, we have developed an experimental strategy to precisely tune cell size over a wide range, allowing us to separately measure the connections among cell geometry, growth, and size, eventually in a variety of organisms and shapes.
Systematic perturbation of cytoskeletal function reveals a linear scaling relationship between cell geometry and fitness.
Monds RD, Lee TK, Colavin A, Ursell T, Quan S, Cooper TF, Huang KC.
Cell Rep. 2014 Nov 20;9(4):1528-37. doi: 10.1016/j.celrep.2014.10.040. Epub 2014 Nov 13.